Citation
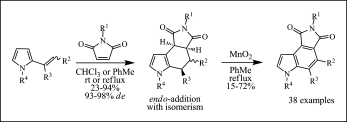
Noland, W. E.; Lanzatella, N. P.; Venkatraman, L.; Anderson, N. F.; Gullickson, G. C. J. Heterocycl. Chem. 2009, 46, 1154.
Publication URL
Publication Year
Image
Publication Number
3
Image
