Citation
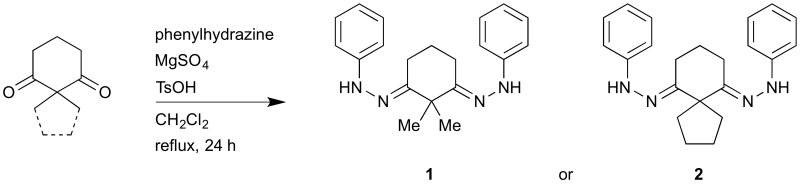
Tritch, K. J.; Capek, J. A.; Klein, N. D.; Noland, W. E. ChemSpider Synthetic Pages 2017, 815, 1.
Publication URL
Publication Year
Image
Publication Number
4
Image
